1.7. Propellants, Pyrotechnics, High explosives
A Calorimetry-based Kinetics Analysis Objective - - To extract chemical kinetics of target energetic materials
- - Tabular form of Arrhenius rate law precisely tracks reaction progress
DSC - - Conversion degree calculation – comparison with DSC experiment
- - Reaction rate calculation – comparison with DSC and one-step kinetics
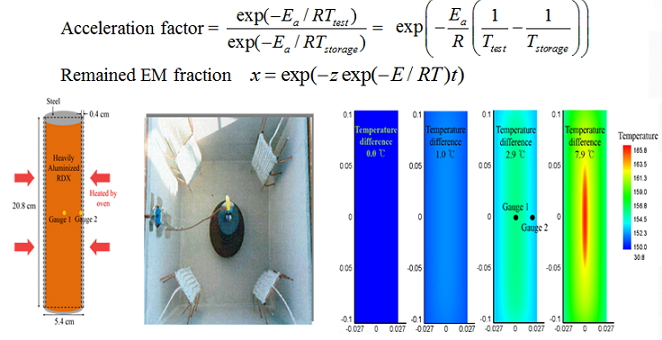 | | Fig. Decomposition experimental | Fig. Simulation |  | | Fig. Aging Experiment | Experimental and Theoretical Study of Pyrotechnic Star Combustion Objective - - Experimental and theoretical value of chromaticity and luminance was carried out
Comparison of experimental & theoretical flame - - Predicting the chromaticity of firework propellant flame was demonstrated
- - The error was 0.2%, 2.5%, 28%, and 0.3% for red, green, blue, and yellow propellant
- - Establishing the technique for luminance measurement
- -The luminance was comparable according to previously reported values
 | | Fig. Experimental Setup | 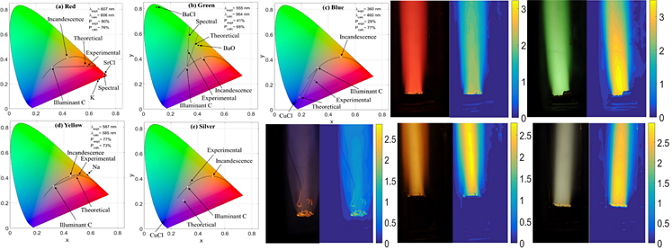 | | Fig. Comparison of experimental and theoretical values | |